
Wartime developments
Switzerland has always depended upon imported fuel, and the outbreak of World War II forced it to consider alternative fuels to keep its vehicles moving. Over two years from 1941 to 1943 the Swiss programme converted around 15,000 vehicles. Constrained by fuel and material availability, it followed these rules:
Private cars below 2 litres engine capacity – dissolved acetylene
Private cars of any size – acetylene generator
Private cars and light commercial vehicles – charcoal gas producer
Heavy commercial vehicles – wood gas producer.
The lack of a fully developed automobile industry forced research and development into the hands of smaller accessory and servicing firms. The Swiss authorities therefore gave them significant support, helping inventors towards success in the compulsory acceptance trails rather than hampering them with unnecessary red tape.
In this Part 1 of the story we’ll talk about acetylene. Switzerland was the only country where this gas was used to any considerable degree. It was experimented with during the Great War, but petrol imports resumed before much progress had been made.
Acetylene
Acetylene is produced by the action of water on calcium carbide, producing acetylene, calcium hydroxide and heat:
CaC2 + 2H20 = C2H2 + Ca(OH)2 + 714 BTU per lb. of calcium carbide.
Calcium carbide is derived from limestone and coal. Switzerland had limestone but had to import its coal. Electric furnaces were needed to produce calcium carbide, and their consumption made this a marginally economical process.
The combustion of a perfect or lean mixture is theoretically very clean, producing CO2 and water with no carbon monoxide or carbon deposits. But acetylene is prone to exothermic dissociation into carbon and hydrogen and to auto-ignition, and not only do rich mixtures disturb cylinder combustion, but the output of heat risks explosion during production in the gas generator – a danger greatly reduced by dissolving the gas in alcohol or water.
Acetylene also has a very high combustion velocity, and spark plug adaptors are needed to resist the propagation of the flame front (see Figure 1).
Figure 1. Plug adaptor for engines operating on acetylene.
The ideal damping liquid to reduce dissociation and auto-ignition was alcohol, but this was rationed in Switzerland during the war and a mix of water and methyl alcohol was found to be the best compromise. This had to be varied during running, because more alcohol was needed at full throttle. Methyl alcohol is poisonous to humans and must be carefully handled. It also dissolves rust and other deposits in fuel pipes – so these must be cleaned regularly.
These considerable disadvantages were not the only ones. Pure acetylene gives only 25 to 30 percent of the power available from a petrol engine before conversion. But damping with petrol, with a petrol consumption of 20 percent of the figure before conversion, gives virtually no power loss, provided the engine is kept free of the carbon deposits caused by insufficient damping.
Precautions
Acetylene-air mixtures are highly flammable and can explode, so some vital precautions were listed for use of acetylene:
Generators required safety valves set at 20 lb. per sq. in.
Generators had to be completely gastight
No iron rods had to be used in cleaning, because they could create a spark
Generators had to be cut out during the last part of any run, to stop the formation of gas
after shutting off the engine (known as post-carburation), to prevent pressure build-up
and escape of gas through safety valves
Cars had ideally to be left in the open for an hour before garaging at night.
Generators
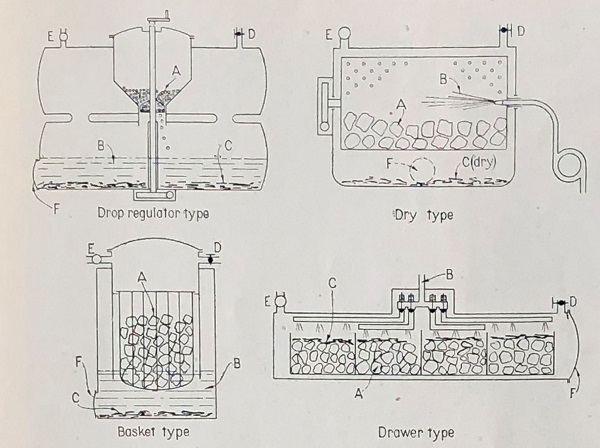
The different types of generator are illustrated in Figure 2.
Figure 2. The four types of acetylene generator.
The drop regulator system enabled the controlled fall of acetylene into water. It had the advantage of preventing post-carburation, because gas production stopped as soon as the car stopped. The basket, drawer and dry production systems were more common, but suffered from post-carburation: so long as water or calcium hydroxide was in the vicinity of carbide, acetylene would continue to be produced and had to be vented through a safety valve. This not only gave an unpleasant odour but risked explosions if the safety valve did not operate correctly.
Despite all these inconveniences, the basket, drawer and dry systems were used, but required considerable safety precautions in their designs.
The drop regulator
This controlled the fall of acetylene into water by rotating perforated elements driven from the dynamo. A pressure valve stopped generation when pressure reached a certain level, and gas production stopped completely when the engine stopped. The Eris system was the only drop regulator type in production at the time. It was the less common system, but was also used with success on several different English cars. Figure 3 shows an installation on a Standard car.
Figure 3. Eris drop-regulation generator on a Standard “Flying Eight”. The leather cover has been removed.
The basket system
This system used a large vertical drum in which a basket was lowered into water. As gas built up, water was displaced into a concentric compensator space and removed water from contact with the carbide held in a basket. Calcium hydroxide sludge had to be removed regularly from the bottom of the drum. Gas was collected in a storage chamber. Figures 4 and 5 show two different installations, both of them awkward and bulky.
Figure 4. Endress basket-type generators. A duplicated installation with external regulation levers.
Figure 5. Riwa basket-type generator with pressure regulation. The generator and gas storage chamber are located on opposite sides to avoid obstructing the luggage space.
The drawer system
This was specifically developed for cars. Separate drawers contained calcium carbide, and in the Carbor system from the Swiss branch of General Motors solenoid-operated valves, remotely controlled from the dashboard, distributed water to each drawer. Figure 6 shows its installation on a small truck, but does illustrate that installation on a car could be not too cumbersome – and could include a front bumper.
Figure 6. G.M. Carbor drawer-type generator with incorporated bumper bar.
Lights on the dashboard indicated the drawer in use, and a pressure gauge showed when one compartment was nearly exhausted and the next was to be taken into use (see Figure 7).
Figure 7. Pressure gauge, control lights and push buttons for a G.M. Carbor installation. Care had been taken to blend the panel into the dashboard.
The system suffered from considerable post-carburation from calcium hydroxide sludge collecting at the top of each basket, so gas was collected in a large container filled with acetone and a porous substance, capable of dissolving a large volume of acetylene and thus increasing gas capacity. Figure 8 shows two of such installations.
Figure 8. On the left, Carba dissolved acetylene bottles housed in the luggage space of a Dodge car. On the right, dissolved acetylene cylinders mounted on a rear panel.
Dry production
This system was technically the most effective. Water was introduced only at the rate needed for the immediate running needs of the car, thus giving a clean chemical reaction with no unwanted residues. Figure 9 shows the Buss system, the most highly developed dry generator at the time, in which a revolving drum with perforated walls contained the carbide. A pressure-operated regulator injected a precisely controlled volume of water through one of the drum pivots, and the rotating drum constantly exposed fresh carbide to water. Calcium hydroxide fell through the drum perforations as a dry powder and collected in a chamber below the drum.
Figure 9. Buss dry-type generator. The drum drive is on the right and the water pump is on the left.
Gas mixing
Mixing of gas with air to supply the engine was by a relatively simple form of carburettor, and the original petrol carburettor was retained to control the flow of damping liquids.
Performance
It was found that the smallest cars suffered virtually no drop in performance, but larger types, because of the resistance of the delivery regulators, lost about 25 per cent of acceleration and 30 per cent of top speed. Even the most powerful cars could reach only 65 to 70 mph. More frequent maintenance was required, since carbon built up in cylinder heads and sludge, damaging to bearings, formed in sumps.
Generators had to be flushed before recharging and at least every 300 to 400 miles – and this was not a job for owner-drivers. Range was between 60 and 100 miles. This made acetylene popular for short journeys in town, for such users as taxi-drivers and doctors.
A curious postscript
All the acetylene systems give an overriding impression of bulk: they fill up the boot; they hang off the front or rear of the car. But our final image, Figure 10, shows that it was just possible to fit the whole lot onto a motorcycle. The Swiss have never been short of ingenuity.
Figure 10. A motorcycle equipped for operation on acetylene.
Part Two of our Technical Talk on alternative fuels in Switzerland during World War II will be on their use of producer gas. Look out for this in a few weeks’ time.
Drawings and photographs courtesy of the Richard Roberts Archive
















Leave a Comment